  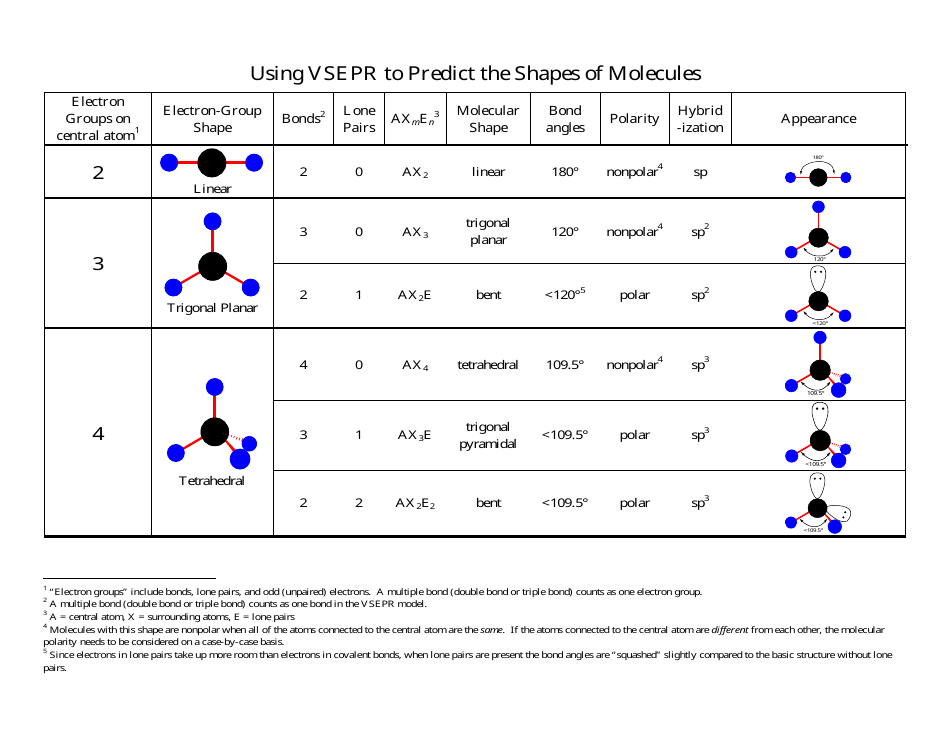
In molecules with more than three atoms, there are many more possible geometries. Thus, the hybridization of both Carbon and Oxygen atoms in the Carbon Monoxide molecule is sp. Iodine pentafluoride (IF 5) is a polar molecule with net 4.81 D. The F-I-F bond angle in IF 5 is 81.9° while the I-F bond lengths are 184.4 pm and 186.9 pm. The central I atom in the IF 5 molecule is sp 3 d 2. Molecules having steric number 2 have sp hybridization. The molecular geometry or shape of IF 5 is square pyramidal while its ideal electron geometry is octahedral. 
Hence Steric Number of Carbon will be: 1+1 2. However, with a triatomic molecule (three atoms), there are two possible geometries: the atoms may lie on a line, producing a linear molecule, or not, producing a bent molecule. For Carbon, we have one lone pair of electrons and one atom ( Oxygen) attached to it. An example of the complexities which arise with polyatomic molecules is molecular geometry: how are the atoms in the molecule arranged with respect to one another? In a diatomic molecule, only a single molecular geometry is possible since the two atoms must lie on a line. A polyatomic molecule contains more than two atoms. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
